Which Of The Following Is A Measure Of Randomness In A System?
Which of the following is a measure of randomness in a system?. Three NADH molecules e. Which simulation allows using the most convenient distribution to communicate opinions about the randomness in measurements. Heat of combustion in kJ mol-1 of benzene at constant pressure will be - R 8314.
B H is the change in entropy the energy available to do work. The combustion of benzene l gives CO_2g and H_2Ol. The higher the entropy of a system the higher the disorder and the higher the unavailability of the systems energy to.
As a measure of randomness or uncertainty the BoltzmannShannon entropy H has become one of the most widely used summary measures of a variety of attributes characteristics in different disciplines. The measure of randomness or disorder in a system is known as. Which of the following statements are NOT true of simulation.
Comparisons between differences in H-values are not valid. The universe favors entropy increases. Six carbon dioxide molecules d.
Every system tries to acquire maximum state of randomness or disorder. C G is the change in free energy. Given that heat of combustion of benzene at constant volume is -32639 kJ mol-1 at 25circ C.
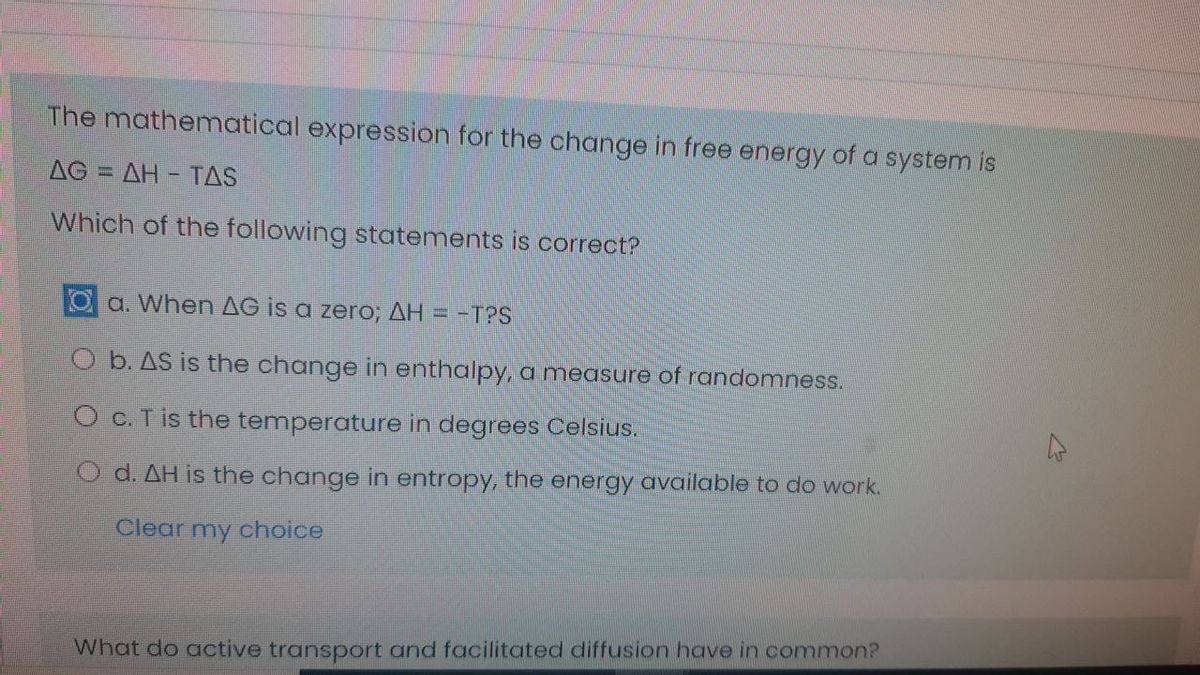
C Entropy is a property of system. The mathematical expression for the change in free energy of a system is G H - TS. D T is the temperature in degrees Celsius.
Entropy is directly proportional to temperature. Bsystem Cexperimentation DNone of above 9.
Entropy is the measurement of randomness of any systemIt is the property of system.
An alternative entropy H K is introduced as a preferred member of a new. Heat of combustion in kJ mol-1 of benzene at constant pressure will be - R 8314. Entropy is a measure of unavailable energy. A ΔS is the change in enthalpy a measure of randomness. Unavailable energy Entropy Temperature The ratio of Entropy of Vaporization and boiling point of a substance remains almost constant. The entropy of the system increases as entropy is considered a measure of randomness of a chemical system. D T is the temperature in degrees Celsius. Three NADH molecules e. Which simulation allows using the most convenient distribution to communicate opinions about the randomness in measurements.
C G is the change in free energy. Which of the following is are correct. The conversion of one molecule of glucose to two pyruvate molecules results in production of. Entropy is a measure of randomness or disorder in a system. If all possible outcomeselements in your sample space have the EXACT same probability of happening than randomness is achieved to its fullest extent. Four ATP molecules b. C ΔG is the change in free energy.





























Post a Comment for "Which Of The Following Is A Measure Of Randomness In A System?"